Breast Cancer Doesn't Just Grow Through Estrogen: The Other Pathways No One Tells Women About
By: Jill Chmielewski, May 15, 2026
Every week, I hear from women who have been told some version of the same sentence:
"Your cancer was ER-positive, so estrogen caused it."
"You can never go near estrogen again."
"Stay on this anti-estrogen drug for the next five, ten, or fifteen years, or your cancer will come back."
I understand the fear behind those statements. I really do. But the more I read the science, the more I see that this story is incomplete. And women deserve the full story.
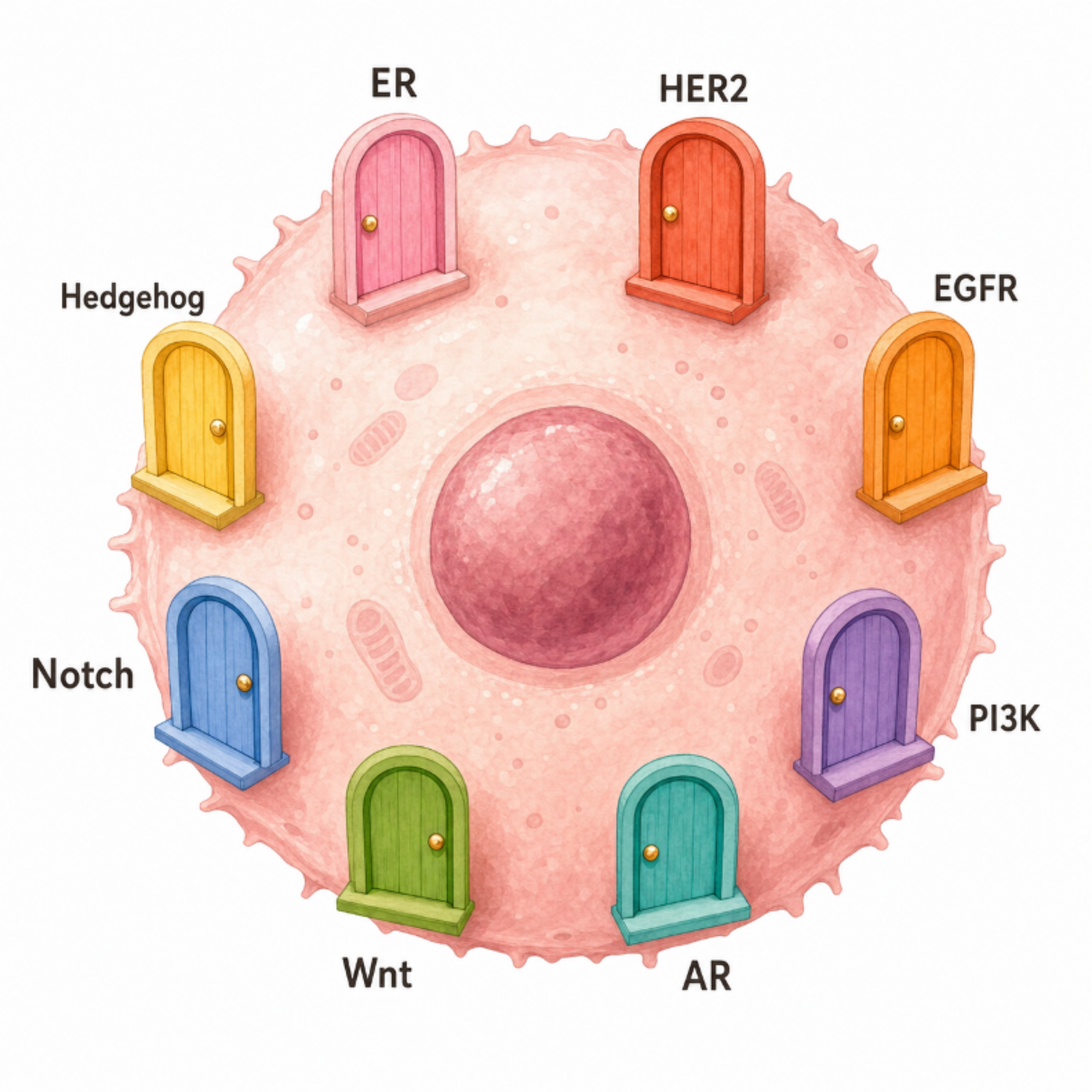
Here is the part most women are never told: breast cancer is not one disease. It is many. And estrogen, while important, is only one of at least ten different pathways a breast cancer cell can use to grow. Even tumors labeled "ER-positive" don't grow only through estrogen. They have backup systems. They have side doors. They have alternate routes. And when one route is blocked, they often switch to another, which is exactly why so many women on long-term estrogen blockers eventually relapse anyway [1].
I want to walk you through these pathways in plain language, organized by cancer type, so you can see what your own diagnosis actually means and what the research really shows.
What "Pathway" Even Means
Let me back up for a minute.
A pathway is just a chain of mini text messages inside a cell. A signal lands on a receptor (think of a receptor as a small antenna on the outside of the cell), and that signal gets passed down a line of proteins until it reaches the nucleus, the cell's control room. The signal tells the cell what to do: grow, divide, repair, survive, die.
Cancer cells are cells that have learned to misuse the same growth pathways your healthy cells use every day. They turn the volume up. They stop listening to "stop" signals. And critically, they have more than one pathway running at the same time.
This is the part that often gets lost in the conversation. When women hear "ER-positive," they often picture a tumor with one button — estrogen, that, if pressed, makes the cancer grow. Press it, cancer grows. Block it, cancer stops.
That is not how cells work. Not even close.
Figure 1. A breast cancer cell with multiple "doors" (signaling pathways). Block one door, and the cell can still use the others.
A Quick Map: Which Pathways Drive Which Type of Cancer
You can see right away that no cancer type runs on just one pathway. And the bottom row is the one that should give every woman pause. I'll come back to it.
If Your Cancer Was ER-Positive (Luminal A or B)
This is the most common type, and probably the most misunderstood. Yes, the estrogen receptor can hear estrogen signals. But it is one of at least six pathways that can keep an ER-positive tumor growing.
1. The estrogen receptor (ER) itself. Estrogen binds to ER inside the cell, the complex moves to the nucleus, and it can turn on genes that drive cell division [3]. This is the pathway tamoxifen and aromatase inhibitors target.
2. PI3K/AKT/mTOR (the "PAM" pathway). About 70% of all breast cancers have some change in this pathway, and around 40% of advanced ER-positive cancers carry a specific PIK3CA mutation that switches it on [4]. This is the main reason endocrine therapy stops working over time. The cancer doesn't need estrogen anymore because it has another engine. This is also why drugs like alpelisib (a PI3K blocker), capivasertib (an AKT blocker), and everolimus (an mTOR blocker) now exist [5].
3. MAPK/ERK. When tamoxifen stops working, this pathway is often why. The cell routes around the blocked ER and grows through MAPK instead [6].
4. Androgen receptor (AR). Here is one most women have never heard of. About 70 to 90% of ER-positive breast cancers also have androgen receptors [7]. And this is important. In ER-positive disease, AR usually acts as a brake, not an accelerator. AR activation can actually push estrogen receptors off the DNA and slow tumor growth. This is why AR-positive tumors tend to have a better prognosis. And why breast surgeon Dr. Rebecca Glaser is uses testosterone in breast cancer patients, and why some researchers are now testing AR agonists (testosterone-like drugs) as a treatment in ER-positive cancer [8].
5. Growth factor receptors (HER family). Even in HER2-negative cancer, the HER family of receptors can remain active in the background, providing signals that promote cell growth [9].
6. Wnt and Notch. These are developmental pathways your body normally uses to grow tissues. In cancer, they get stuck "on" and contribute to growth, resistance to treatment, and the survival of cancer stem cells [10].
So when a woman is told that her ER-positive cancer was "caused by estrogen," that is a simplification of a simplification. Her tumor likely had at least three or four pathways running before anyone even ran a biopsy.
If Your Cancer Was HER2-Positive
HER2 is a receptor tyrosine kinase. That is a fancy name for a receptor on the outside of the cell that, when activated, fires off a strong "grow and divide" signal [11].
In HER2-positive cancers, the cells have extra copies of HER2, sometimes 50 to 100 times the normal amount. That is the main growth driver. But HER2 doesn't act alone.
1. HER2 itself. Targeted by trastuzumab, pertuzumab, and the newer HER2 tyrosine kinase inhibitors [12].
2. PI3K/AKT/mTOR. When trastuzumab stops working, PI3K activation is one of the main escape routes [12]. This is why some women now receive trastuzumab combined with an mTOR inhibitor.
3. MAPK/ERK. Another backup proliferation pathway.
4. AR (androgen receptor) + Wnt feedback loop. In HER2-positive tumors, AR can actually team up with HER2 through a loop involving Wnt signaling and HER3, which keeps the tumor alive [13]. This is one reason HER2-positive tumors that also express AR sometimes have worse outcomes than ER-positive AR-positive tumors.
5. ER (when present). Many HER2-positive cancers are also ER-positive ("triple positive"). The two pathways talk to each other and trade signals back and forth, which is why oncologists often recommend both anti-HER2 and anti-estrogen therapy at the same time [14].
If Your Cancer Was Triple Negative (TNBC)
Triple negative cancers have no estrogen receptor, no progesterone receptor, and no HER2.
So if estrogen were the cause, these cancers should not exist. But they do. And they are often the most aggressive type.
TNBC grows through a whole different set of pathways:
1. EGFR (HER1). The first cousin of HER2. A major growth driver in TNBC [15].
2. PI3K/AKT/mTOR. Frequently activated, often through loss of a brake protein called PTEN [16].
3. Wnt/β-catenin. Drives proliferation, stem cell maintenance, and the spread of cancer to other organs [17].
4. Notch. Especially Notch1 and Notch4. Notch4 keeps a quiet, sleeping population of cancer stem cells alive that resist chemo [18].
5. Hedgehog. Another developmental pathway hijacked to feed stemness and survival [15].
6. JAK/STAT3. Constantly switched on in TNBC and inflammatory breast cancer. It cross-talks with Wnt, Notch, and EGFR [19].
7. Hippo/YAP-TAZ. A newer player. Drives size, survival, and stemness [20].
8. TGF-β. Starts off as a tumor brake, then flips to become a metastasis driver as the cancer progresses [15].
9. NF-κB. Inflammation and survival signaling. Closely tied to stress, infection, and metabolic disease [21].
10. AR (androgen receptor). In about 10–43% of TNBC cases, AR is expressed. In a subset called "LAR" (luminal androgen receptor) TNBC, AR actually drives growth by pushing EGFR signaling [22].
That is at least ten growth pathways. None of which is estrogen.
Figure 2. When the ER lane is blocked, the cell can switch lanes — PI3K, MAPK, Wnt, Notch, AR — and keep growing.
And Then There Are Cancer Stem Cells
This is the section I want every woman with a breast cancer history to read twice.
Inside every tumor, there is a small population of cells called cancer stem cells, or breast cancer stem cells (BCSCs). They are the "seeds." They are the cells most responsible for recurrence and metastasis, the cells that survive chemo, the cells that wake up years or decades later [23].
And here is what almost no one tells women:
Cancer stem cells often have very low or no estrogen receptors at all [24].
Read that again.
The "seed" cells of even an ER-positive tumor are frequently ER-negative. They use Notch, Wnt, Hedgehog, JAK/STAT3, and PI3K/AKT/mTOR to stay alive [23, 25]. They do not respond to tamoxifen or aromatase inhibitors the way the bulk of the tumor does.
In fact, in some cases, anti-estrogen (endocrine) therapy actually expands this stem cell population by pushing them into a quiet, dormant state that hides from treatment [24, 25].
So a woman on tamoxifen or an aromatase inhibitor is doing something for the bulk of the tumor cells. She is doing very little, and sometimes the opposite of what she thinks for the seed cells.
That’s why it is so important to look at the true absolute risk reduction benefits when making decisions about endocrine therapy and other cancer treatments.
The High-Dose Estrogen Paradox
This is the part that breaks most women's brains. It broke mine when I first read it.
We know from over 50 years of research that high-dose estrogen can actually shrink some breast cancers [26].
Back in the 1940s and 1950s, before tamoxifen existed, advanced breast cancer was often treated with high-dose estrogen. It worked. Tumors regressed. Women lived longer. Then tamoxifen came along, and the field forgot.
In 2009, Dr. Matthew Ellis and colleagues conducted a randomized trial at Washington University, which was published in JAMA. They gave 6 mg or 30 mg of estradiol per day to postmenopausal women with ER-positive metastatic breast cancer who had stopped responding to aromatase inhibitors [26]. Roughly 30% of the women achieved clinical benefit, including some with complete or partial tumor regression [26]. The lower 6 mg dose was just as effective as the 30 mg dose, with fewer side effects.
How can the same hormone that some say "feeds" cancer also shrink it?
The answer is biology. Cells that have been starved of estrogen for years adapt by becoming hyper-sensitive to estrogen. When estrogen finally returns, the cells become overwhelmed and trigger apoptosis, a programmed form of cell death [27]. It's a stress response, not a feeding response.
Dr. Zsuzsanna Suba, a researcher whose work I have followed closely, has written extensively about this paradox. Her position is that estrogen, far from being the villain, is actually a regulatory signal that helps cells repair DNA damage and stay genomically stable [28]. When estrogen signaling is disrupted through aging, menopause, BRCA mutations, or long-term suppression, the body loses one of its most important systems for keeping cells in line.
If estrogen itself caused cancer, women would develop breast cancer every month of their reproductive lives, when estrogen levels are highest. They don't. Pregnancy, the highest estrogen state of a woman's life, actually lowers lifetime breast cancer risk [28]. Something more complicated is going on.
Estrogen blockade may be the right choice for a limited period of time. But women deserve to know that the story is more nuanced than they have been told.
What This Means for the Women Reading This
A few takeaways I keep coming back to:
Breast cancer is not one disease. It is many. And estrogen is potentially one driver among many, never the only one, and often not even the most important one.
"Endocrine resistance" usually means the tumor switched lanes. When tamoxifen or an aromatase inhibitor stops working, it is rarely because estrogen "got past" the drug. It is because the tumor activated another pathway, most often PI3K/AKT/mTOR or MAPK [4, 6]. The block on estrogen still works. The tumor just doesn't need estrogen anymore.
Triple-negative breast cancer exists, and it grows without any estrogen at all. If estrogen caused breast cancer, this type of cancer would be impossible. It isn't. It's actually the hardest to treat.
Cancer stem cells are often estrogen-receptor negative [24]. The "seeds" of recurrence may not respond to anti-estrogen therapy the way women are told they will.
High-dose estrogen has been used to treat metastatic breast cancer for over 50 years [26]. The dominant narrative has not caught up with the data.
Women deserve to know that "ER-positive" is the start of the conversation, not the end of it. They deserve a benefits-and-risks discussion that includes the risks of not using hormones — bone loss, muscle loss, heart disease, brain fog, cognitive decline, and sexual dysfunction. They deserve clinicians who understand that hormones are not on/off switches for cancer. They are regulatory messengers that the body needs for repair, signaling, and balance.
We can do better. The science already supports it.
A Word on Repurposed Drugs
There is also a growing body of work looking at older, well-studied drugs that may quietly target some of these same pathways. Dr. Paul Marik and the Independent Medical Alliance have pulled together a Cancer Care guide that examines medications such as metformin, mebendazole, ivermectin, doxycycline, and propranolol, alongside nutraceuticals such as curcumin, resveratrol, and vitamin D, as possible adjuncts to standard cancer care. The idea isn't to replace conventional treatment. It's to recognize that cancer is a metabolic and multi-pathway disease, and that blocking only one lane may not be enough.
This is still an emerging space, and the evidence varies from drug to drug. But it's worth knowing about, especially if you're a woman who wants to ask thoughtful questions about every layer of a treatment plan. I'm not recommending any specific protocol here. I'm pointing to a conversation worth having with a clinician who is willing to look at the whole picture.
Links: https://imahealth.org/research/cancer-care/ | https://imahealth.org/wp-content/uploads/2025/02/approach-to-repurposed-drugs-for-cancer.pdf
References
1. du Rusquec, P., Blonz, C., Frenel, J. S., & Campone, M. (2020). Targeting the PI3K/Akt/mTOR pathway in estrogen-receptor positive HER2 negative advanced breast cancer. Therapeutic Advances in Medical Oncology, 12, 1758835920940939. https://doi.org/10.1177/1758835920940939
2. Almansour, N. M. (2022). Triple-Negative Breast Cancer: A Brief Review About Epidemiology, Risk Factors, Signaling Pathways, Treatment and Role of Artificial Intelligence. Frontiers in Molecular Biosciences, 9, 836417. https://doi.org/10.3389/fmolb.2022.836417
3. Khan, M. Z. I., Uzair, M., Nazli, A., & Chen, J. Z. (2022). An overview on Estrogen receptors signaling and its ligands in breast cancer. European Journal of Medicinal Chemistry, 241, 114658. https://doi.org/10.1016/j.ejmech.2022.114658
4. Liu, L., Graff, S. L., & Wang, Y. (2024). New Emerging Therapies Targeting PI3K/AKT/mTOR/PTEN Pathway in Hormonal Receptor-Positive and HER2-Negative Breast Cancer—Current State and Molecular Pathology Perspective. Cancers, 17(1), 16. https://doi.org/10.3390/cancers17010016
5. Wali, A. F., Talath, S., El Tanani, M., Rashid Rangraze, I., Babiker, R., Shafi, S., & Bansal, R. (2025). PI3K/AKT/mTOR Pathway in Breast Cancer Pathogenesis and Therapy: Insights into Phytochemical-Based Therapeutics. Nutrition and Cancer, 77(9), 938–958. https://doi.org/10.1080/01635581.2025.2521884
6. Javankiani, S., Bolandi, S., Soleimani, A., Meigoli, M., Parsafar, M., Safaei, S., et al. (2025). MAPK signaling mediates tamoxifen resistance in estrogen receptor-positive breast cancer. Molecular and Cellular Biochemistry, 480(9), 4973–4989. https://doi.org/10.1007/s11010-025-05304-0
7. Dai, C., & Ellisen, L. W. (2023). Revisiting Androgen Receptor Signaling in Breast Cancer. The Oncologist, 28(5), 383–391. https://doi.org/10.1093/oncolo/oyad049
8. Asemota, S., Effah, W., Holt, J., Johnson, D., Cripe, L., Ponnusamy, S., et al. (2024). A molecular switch from tumor suppressor to oncogene in ER+ breast cancer: Role of androgen receptor, JAK-STAT, and lineage plasticity. Proceedings of the National Academy of Sciences, 121(40), e2406837121. https://doi.org/10.1073/pnas.2406837121
9. Hayashi, N., Iwamoto, T., Qi, Y., Niikura, N., Santarpia, L., Yamauchi, H., Nakamura, S., Hortobágyi, G. N., Pusztai, L., Symmans, W. F., & Ueno, N. T. (2017). Bone metastasis-related signaling pathways in breast cancers stratified by estrogen receptor status. Journal of Cancer, 8(6), 1045–1052. https://doi.org/10.7150/jca.13690
10. Mohamed, S., Badawy, T., Hussein, M., Salama, M., El Kerm, Y., & Mohsen, M. (2025). The involvement of exosomes in the tumorigenicity of breast cancer cell lines through the crosstalk between STAT3, Notch, and Wnt signaling pathways. Discover Oncology, 16(1). https://doi.org/10.1007/s12672-025-03334-0
11. Fujimoto, Y., Morita, T. Y., Ohashi, A., Haeno, H., Hakozaki, Y., Fujii, M., Kashima, Y., Kobayashi, S. S., & Mukohara, T. (2020). Combination treatment with a PI3K/Akt/mTOR pathway inhibitor overcomes resistance to anti-HER2 therapy in PIK3CA-mutant HER2-positive breast cancer cells. Scientific Reports, 10, 21762. https://doi.org/10.1038/s41598-020-78646-y
12. Li, Q., Lv, D., Sun, X., Wang, M., Cai, L., Liu, F., Li, C., Zhao, J., Sun, J., Shi, Y., & Ma, F. (2024). Inetetamab combined with sirolimus and chemotherapy for the treatment of HER2-positive metastatic breast cancer patients with abnormal activation of the PI3K/Akt/mTOR pathway after trastuzumab treatment. Cancer Innovation, 3, e145. https://doi.org/10.1002/cai2.145
13. Ni, M., Chen, Y., Lim, E., Wimberly, H., Bailey, S. T., Imai, Y., Rimm, D. L., Liu, X. S., & Brown, M. (2011). Targeting Androgen Receptor in Estrogen Receptor-Negative Breast Cancer. Cancer Cell, 20(1), 119–131. https://doi.org/10.1016/j.ccr.2011.05.026
14. Xie, J., Yang, Z., Li, Z., Zhang, T., Chen, H., Chen, X., Dai, Z., Chen, T., & Hou, J. (2025). Triple-positive breast cancer: navigating heterogeneity and advancing multimodal therapies for improving patient outcomes. Cancer Cell International, 25(1). https://doi.org/10.1186/s12935-025-03680-7
15. Medina, M. A., Oza, G., Sharma, A., Arriaga, L. G., Hernández Hernández, J. M., Rotello, V. M., & Ramirez, J. T. (2020). Triple-Negative Breast Cancer: A Review of Conventional and Advanced Therapeutic Strategies. International Journal of Environmental Research and Public Health, 17(6), 2078. https://doi.org/10.3390/ijerph17062078
16. Sirek, T., Król-Jatręga, K., Borawski, P., Zmarźly, N., Boroń, D., Ossowski, P., Nowotny-Czupryna, O., Boroń, K., Janiszewska-Bil, D., Mitka-Krysiak, E., & Grabarek, B. O. (2025). Distinct mRNA expression profiles and miRNA regulators of the PI3K/AKT/mTOR pathway in breast cancer: insights into tumor progression and therapeutic targets. Frontiers in Oncology, 14, 1515387. https://doi.org/10.3389/fonc.2024.1515387
17. Stojanovic, L., Tripathi, K., Gohari, Z., Rutherford, J. L., Rajendran, S., Metcalfe, T. X., Zhang, S., Baylin, S. B., Topper, M., Nephew, K. P., & Rassool, F. V. (2026). DNMTi in combination with PARPi inhibits aberrant Wnt/β-catenin signaling and tenascin-C pathways, cancer stemness and metastasis in triple negative breast cancer [Abstract]. Cancer Research, 86(7 Suppl), Abstract 2245. https://doi.org/10.1158/1538-7445.am2026-2245
18. Zhou, L., Wang, D., Sheng, D., Xu, J., Chen, W., Qin, Y., Du, R., Yang, X., He, X., Xie, N., Liu, S., & Zhang, L. (2020). NOTCH4 maintains quiescent mesenchymal-like breast cancer stem cells via transcriptionally activating SLUG and GAS1 in triple-negative breast cancer. Theranostics, 10(5), 2405–2421. https://doi.org/10.7150/thno.38875
19. Ibrahim, S. A., Gadalla, R., El-Ghonaimy, E. A., Samir, O., Mohamed, H. T., Hassan, H., Greve, B., El-Shinawi, M., Mohamed, M. M., & Götte, M. (2017). Syndecan-1 is a novel molecular marker for triple negative inflammatory breast cancer and modulates the cancer stem cell phenotype via the IL-6/STAT3, Notch and EGFR signaling pathways. Molecular Cancer, 16(1), 57. https://doi.org/10.1186/s12943-017-0621-z
20. Xu, T., Liu, K., Zhang, Y., Chen, Y., & Yin, D. (2024). EGFR and Hippo signaling pathways are involved in organophosphate esters–induced proliferation and migration of triple-negative breast cancer cells. Environmental Science and Pollution Research, 31(29), 41939–41952. https://doi.org/10.1007/s11356-024-33872-z
21. Poyonov, M. M. U., Bui, A. T. N., Lee, S.-Y., Lee, G.-H., & Jeong, H.-G. (2025). ERα36 Promotes MDR1-Mediated Adriamycin Resistance via Non-Genomic Signaling in Triple-Negative Breast Cancer. International Journal of Molecular Sciences, 26(15), 7200. https://doi.org/10.3390/ijms26157200
22. Fang, T., Hu, S., Song, X., Wang, J., Zuo, R., Yun, S., Jiang, S., & Guo, D. (2024). Combination of monensin and erlotinib synergistically inhibited the growth and cancer stem cell properties of triple-negative breast cancer by simultaneously inhibiting EGFR and PI3K signaling pathways. Breast Cancer Research and Treatment, 207(2), 435–451. https://doi.org/10.1007/s10549-024-07374-y
23. Jan, A., Sofi, S., Jan, N., & Mir, M. A. (2025). An update on cancer stem cell survival pathways involved in chemoresistance in triple-negative breast cancer. Future Oncology, 21(6), 715–735. https://doi.org/10.1080/14796694.2025.2461443
24. Ma, R., Karthik, G.-M., Lövrot, J., Haglund, F., Rosin, G., Katchy, A., Zhang, X., Viberg, L., Frisell, J., Williams, C., Linder, S., Fredriksson, I., & Hartman, J. (2017). Estrogen receptor β as a therapeutic target in breast cancer stem cells. JNCI: Journal of the National Cancer Institute, 109(3), djw236. https://doi.org/10.1093/jnci/djw236
25. Rodriguez, D., Ramkairsingh, M., Lin, X., Kapoor, A., Major, P., & Tang, D. (2019). The Central Contributions of Breast Cancer Stem Cells in Developing Resistance to Endocrine Therapy in Estrogen Receptor (ER)-Positive Breast Cancer. Cancers, 11(7), 1028. https://doi.org/10.3390/cancers11071028
26. Ellis, M. J., Gao, F., Dehdashti, F., Jeffe, D. B., Marcom, P. K., Carey, L. A., Dickler, M. N., Silverman, P., Fleming, G. F., Kommareddy, A., Jamalabadi-Majidi, S., Crowder, R., & Siegel, B. A. (2009). Lower-Dose vs High-Dose Oral Estradiol Therapy of Hormone Receptor–Positive, Aromatase Inhibitor–Resistant Advanced Breast Cancer: A Phase 2 Randomized Study. JAMA, 302(7), 774–780. https://doi.org/10.1001/jama.2009.1204
27. Sarmiento-Castro, A., Caamaño-Gutiérrez, E., Sims, A. H., Hull, N. J., James, M. I., Santiago-Gómez, A., Eyre, R., Clark, C., Brown, M. E., Brooks, M. D., Wicha, M. S., Howell, S. J., Clarke, R. B., & Simões, B. M. (2020). Increased Expression of Interleukin-1 Receptor Characterizes Anti-estrogen-Resistant ALDH+ Breast Cancer Stem Cells. Stem Cell Reports, 15(2), 307–316. https://doi.org/10.1016/j.stemcr.2020.06.020
28. Suba, Z. (2024). Estrogen Regulated Genes Compel Apoptosis in Breast Cancer Cells, Whilst Stimulate Antitumor Activity in Peritumoral Immune Cells in a Janus-Faced Manner. Current Oncology, 31(9), 4885–4907. https://doi.org/10.3390/curroncol31090362
For more information on this topic, see my other articles:
Hormones & Breast Cancer: What’s Fact, What’s Fiction?
The History of Breast Cancer Treatments and the Misguided War on Estrogen
The Truth About Estrogen, Breast Cancer, and Hormone Therapy: What the Latest Research Reveals
Disclaimer: This article is for educational and informational purposes only and is not intended to replace personalized medical advice or individualized care. It is meant to help you understand your physiology, explore evidence-based options, and make informed choices about your health and wellness. Healthcare should be a partnership, not a permission slip, and proactive care is just as essential as treatment. Use this information to engage in open, collaborative discussions with your provider or to make empowered decisions that align with your own values, goals, and comfort level. You are the ultimate authority on your body.